- Home/
- Covid-Tests/
- 10 Questions About At-Home COVID-19 Tests That You Were Too Afraid To Ask
10 Questions About At-Home COVID-19 Tests That You Were Too Afraid To Ask
January 12, 2023
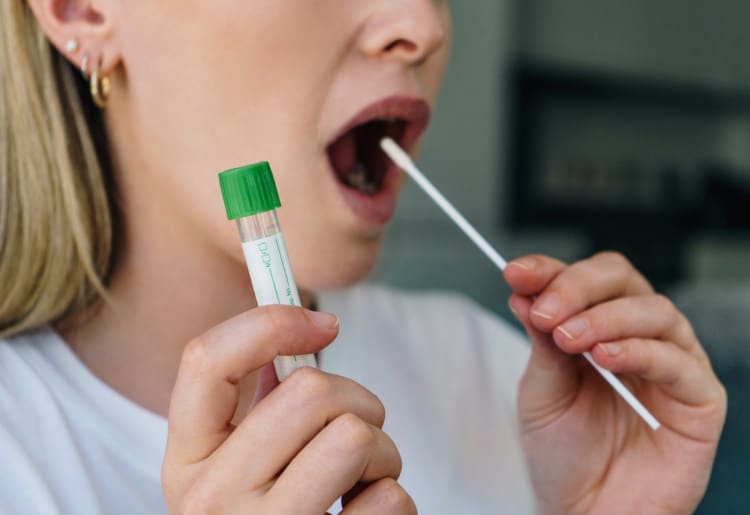
January 12, 2023

Access to onsite testing, however, hasn’t been easy for everyone. Now, the US Food and Drug Administration has issued an Emergency Use Authorization for a select few diagnostic companies to create and sell at-home COVID-19 tests.
So how do at-home COVID-19 test kits work, and how legitimate are they? Let’s take a look at some FAQs and explore the facts surrounding them.
At-home COVID-19 tests range in price from around $109 (Everlywell) to $155. Most diagnostic companies require upfront payment for test kits, but will send recipients an itemized receipt of services after test results have been analyzed. This means you can then submit the itemized receipt and any other necessary information through your health insurance provider’s claims submission process for possible reimbursement.
Pixel by LabCorp, on the other hand, does not require upfront payment for test kits. Instead, the company seeks reimbursement from health insurance providers, collects payment via credit card, or utilizes government funds if the person who tests is uninsured.
Currently, the FDA has authorized the sale of at-home coronavirus tests by only a select few diagnostic companies. Those companies obtained the rights to sell at-home COVID-19 tests under the FDA’s Emergency Use Authorization policy. It’s important to note, however, that receiving an EUA does not automatically mean the product is FDA-approved. Rather, the EUA authorization grants permission to select companies to deliver unapproved products during a public health crisis such as the COVID-19 pandemic.
When the FDA first authorized these companies to distribute at-home test kits under the EUA, the organization stated they worked closely with LabCorb, a global company that provides diagnostic services. According to the FDA, this ensures results collected from at-home test kits are as accurate as those administered in a doctor’s office, hospital, or other testing facility.
Since diagnostic companies providing at-home COVID-19 testing kits are not healthcare providers, most cannot bill health insurances directly. The exception is Pixel by LabCorp, which seeks reimbursement from healthcare providers or government funds, or collects payment via debit or credit card.
Companies such as Everlywell that offer at-home lab tests will send a receipt containing information your insurance provider needs to reimburse costs. After your tests results come back, you can download the necessary information and submit the itemized receipt through your health insurance provider’s claims process.
Once deemed eligible, you or your doctor can order your at-home test kit to receive overnight or within 3 business days. Depending on the test you’ve chosen, you will follow instructions for either a nasal swab, throat swab, or saliva-based test. You can drop your completed sample in a drop box near you and expect test results within 24 to 72 hours after the lab has received it.
Molecular (RT-PCR) at-home COVID-19 tests work by detecting the virus’s genetic material within an infected person’s body. If the lab that receives your test finds coronavirus genetic material in your sample, you will receive a positive result—and you’ll be notified shortly after.
You should hear back about results within 24 to 72 hours of the lab receiving your sample. You will be notified via email or another form of communication you provide when filling out the online information. Additionally, many companies send along a prepaid UPS or FedEx shipping label to make the process of getting results back to the lab as efficient as possible.
Yes. Even if you have little to no symptoms of COVID-19, you can still be infectious. Most people carrying the COVID-19 virus go through a pre-symptomatic phase (usually the first 7 days after exposure) in which they are infectious but display no symptoms. Those individuals may or may not go on to develop symptoms of COVID-19, but they are more likely to spread COVID-19 in the 48 hours prior to the beginning of their symptoms.
If you live alone, make sure to stay at home and away from friends, family, pets, and public settings. If you live with others, choose a room to self-isolate in. Other ways to protect those you live with are:
At-home COVID-19 test kits test for active COVID-19 infection—meaning if you suspect you currently have COVID-19 and wish to test at home, these tests are for you. They are generally collected via nasal swab, throat swab, or a tube of saliva.
Antibody kits, on the other hand, are more suitable for those who suspect they may have already had COVID-19 and have begun to recover, or build antibodies. Antibody tests, such as those offered by HealthLabs and Health Testing Centers, test for a type of antibody called immunoglobulin G (IgG). If you’ve been infected and have recovered within the past few weeks or months, your immune system may have produced detectable IgG antibodies to fight off the illness.
Unlike at-home COVID-19 tests, antibody tests may require blood samples from a finger prick or blood draw administered by a nearby laboratory collection site.
Currently, all at-home COVID-19 test kits authorized by the FDA are molecular tests administered using a nasal swab, throat swab, or saliva. According to the FDA, results from molecular tests are highly accurate and do not need to be repeated, unlike a negative result from an antigen test.
Most diagnostic companies claim their at-home COVID-19 tests offer a sensitivity of at least 98 percent or higher—a solid chance of catching active infections if present.
Companies like Everlywell work with independent board certified physicians to review test results. A telehealth provider will also contact you if your test is positive.
Additionally, the company discloses that, by law, all positive and negative results will be reported to appropriate state public health authorities. Outside of that, your test results will not be shared with or accessible to anyone.
Several of the diagnostic companies authorized by the FDA to distribute at-home COVID-19 tests require you to take an online questionnaire or have physician authorization to be eligible. However, a few tests, such as the saliva test offered by Vault Health, require you to do little more than answer a few demographic questions prior to purchase.
Even after a vaccine has been released, many experts agree that we won’t see a future without masks anytime soon. Accessible, efficient at-home testing is an integral part of learning to live with the COVID-19 virus and identifying outbreaks as they start to occur.
Brittany Dick writes for Top10.com and is a once full-time, professional communicator turned freelance writer who specializes in crafting accurate health, wellness, and nutrition information. Since entering the freelance realm, Brittany has helped healthcare professionals tell the most impactful stories of their brand and deliver valuable health education to online audiences across the country.